Contents
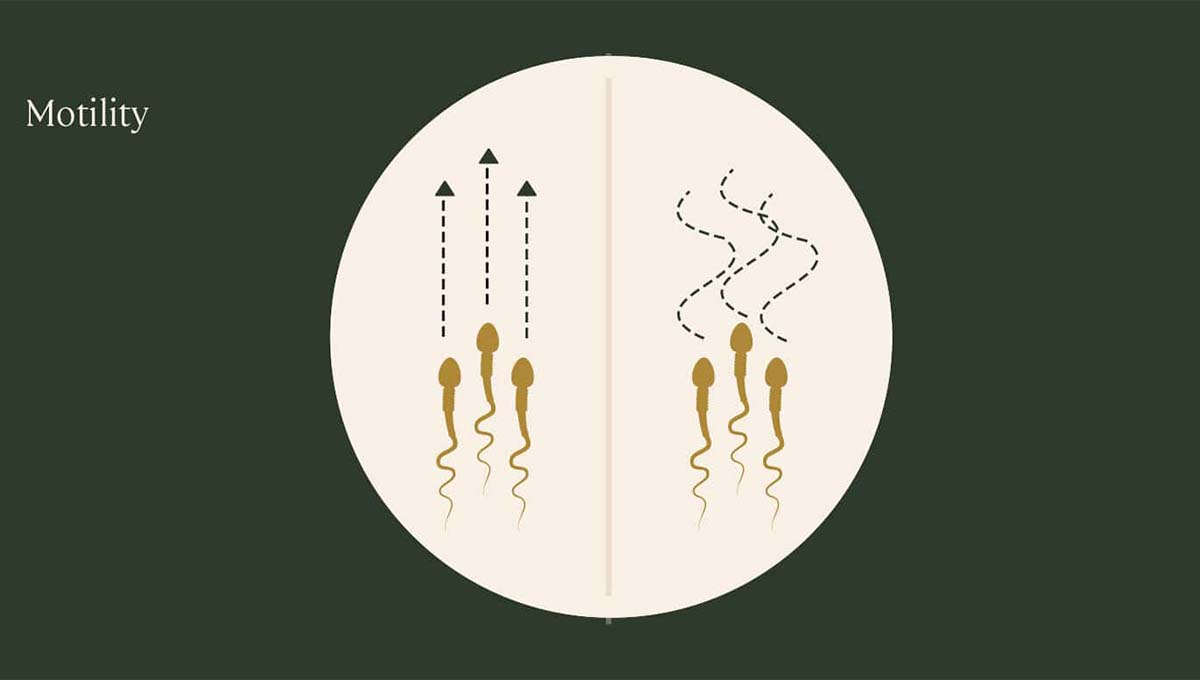
What is sperm motility?
Sperm motility means sperm movement. It is a very important factor for male fertility. Low sperm motility, which Doctors also call “asthenospermia” is therefore characterised by insufficient forward sperm movement.
If sperm is unable to effectively move through the cervix and towards the fallopian tubes then it cannot facilitate egg fertilisation. This can make it difficult for men to naturally father a child.
However, there are some measures men can take to enhance their sperm motility.
Example: diagnosing low sperm motility
www.amitamin.com/en/fertilsan-m New life deserves the best possible start!We provide the essential building blocks for this.
Male fertility testing examines three main factors that affect reproductive capacity.
During a semen analysis, fertility specialists will assess sperm count (sperm concentration within a sample), morphology (the percentage of “normal” forms), and motility percentage.
A man is classified as fertile by the WHO (World Health Organisation), when he has a minimum of 32% Grade A + Grade B (see below) motility.
If, for example. Grade A “highly progressive motile” sperm was 12% and Grade B “progressive motile” sperm was 21%, the sum is 21% + 12% = 33%. The male individual would therefore be over the 32% minimum and therefore be classified as having sufficiently motile sperm.
Keep in mind that sperm count (WHO minimum is 20 million / ml) and sperm morphology / % normal forms (WHO minimum is 4%) both need to be above the WHO minimum thresholds for the male to be classified as fertile.
Sperm motility classification
There are different classifications of sperm motility.
Grade A sperm are the strongest and move quickly in a straight line.
Grade B sperm still move forward quickly, however they tend to travel in a non-direct path.
Grade C sperm are motile, but not progressive. Their tails vibrate but the sperm are unable to move forward.
The final classification, Grade D, refers to sperm that are completely immotile and cannot move.
Men with less than 32% Grade A and Grade B sperm motility are considered to have low sperm motility and are called “subfertile”. In this situation, men need to look at making lifestyle changes to help improve their sperm motility and overall reproductive health if they wish to start a family.
What does Grade A and Grade B motility look like?
The below video nicely demonstrates the differences in forward movement
The sperm cells with Grade A motility move fast and in a straight line. These cells are visible between 0:03 – 0:09 seconds on the below video. The Grade A motility cells move fast from the bottom left to the top right hand side of the screen.
Sperm cells with Motility Grade B are very visible in the second short clip between 0:16 – 0:20. Grade B cells make up the majority of the cells in the upper half of the screen. There are also some very fast moving Grade A cells visible in the lower half of the screen.
What causes low sperm motility?
There is a multitude of causes of poor sperm motility. Some of these include
- Stress: When the body is stressed, the excess Cortisol in the body and resulting oxidative stress can affect sperm production and the health of spermatozoa. Relieving stress can help to improve sperm motility. Effective strategies are regular exercise (preferably low impact sports like swimming, jogging, rowing or cycling), meditation (e.g. check out MBSR – google is your friend) and yoga. Read our article on fertility yoga.
- Alcohol: Sperm production and development can be adversely affected by excessive alcohol consumption1 La Vignera S, Condorelli RA, Balercia G, Vicari E, Calogero AE. Does alcohol have any effect on male reproductive function? A review of literature. Asian Journal of Andrology. 2013. 15(2): 221-5, decreasing motility and overall sperm health.
- Heat: Sperm cells are highly sensitive to heat. This is of course the reason, why the testicles are located outside of the main body. If exposed to high temperatures for prolonged periods sperm production will suffer, which can lead to a decrease in sperm concentration and motility. Aspiring fathers should not spend a long time in saunas, hot-tubs, or other environments with excessive temperatures. Loosely fitting underwear can help to keep sperm at the optimal temperature. Also avoid using laptops directly on the lap for extended periods.
- Sex: Excessive sex can affect overall sperm count and motility. For men trying to father a child it”s recommended to only have sex every two-three days in the lead-up to ovulation. This will allow for healthy, viable sperm to develop. During ovulation the frequency of sex can increase to help improve conception chances.
- Poor nutrition: The body requires certain vitamins and minerals to support healthy sperm motility. A nutrient deficient diet can adversely affect sperm health. Conversely, consuming the right nutrients has been found to boost sperm motility.
- Heavy metals: Exposure to heavy metals through cheap processed foods can reduce sperm health and cause abnormalities. The solution is stop eating fast food, cook more at home and eat more organic vegetables.
- Smoking: Tobacco smoke has dozens harmful chemicals that can reduce sperm motility and viability. If you are trying to start a family, stop smoking!
- Bluetooth: Two clinical studies have shown that the now almost ubiquitous Bluetooth radiation from electronic devices reduces sperm motility and viability2 Agarwal A, Desai NR, Makker K, Varghese A, Mouradi R, Sabanegh E, Sharma R. Effects of radiofrequency electromagnetic waves (RF-EMW) from cellular phones on human ejaculated semen: an in vitro pilot study. Fertility and Sterility. 2009. 92(4): 1318-25, 3 Gutschi T, Mohamad Al-Ali B, Shamloul R, Pummer K, Trummer H. Impact of cell phone use on men’s semen parameters. Andrologia. 2011. 43(5): 312-6. So switch off the bluetooth and get those cables back out!
- Genetics: A genetic condition causing DNA fragmentation can lead to poor sperm motility. This condition is often hereditary.
- Recreational drugs: Clinical studies have confirmed that anabolic steroids and marijuana do significant damage to sperm quality. If you intend to father a child, you should stop consuming such substances immediately.
- Subscription drugs: if you are taking subscription medication, please check their potential impact with your pharmacist and / or GBP or physician.
How is sperm motility tested?
As part of a semen analysis sperm cells are counted under the microscope in a counting chamber using a grid called hemacytometer.
Treatment options for low sperm motility
There are several way to treat “suboptimal” sperm motility, depending on the severity:
- Men with Grade A + B motility around 30% can often increase their sperm quality with lifestyle changes like low impact regular exercise and a cleaner diet with a focus on the right foods and supplements. This needs to be done for a minimum of three months until a pregnancy has been achieved.
- Men with Grade A + B motility around 20% can also improve their readings with lifestyle changes and supplements until conception. Depending on the couple’s health history and age, artificial fertilisation techniques like intrauterine insemination (IUI), IVF or ICSI may be recommended. Of these IUI is the cheapest treatment, but it also has the lowest success rates (10-20% of couples)
- Men with Grade A + B motility 15% or less: A urologist or male fertility expert evaluates whether a correctable medical condition like varicocele can be diagnosed and treated. Again, these men can also improve their readings with lifestyle changes and supplements, however the most successful treatment option for these men is IVF in combination with ICSI.
Which nutrients are proven to boost motility?
Making positive lifestyle changes can significantly help men with fertility problems. In particular, improved nutrition is a big advantage.
There are certain nutrients that can assist to improve sperm motility.
Studies have found:
- 2 g/day of the amino acid L-carnitine together with vitamin E has been shown to increase sperm motility by 59% over a period of eight months4 Lenzi A, Lombardo F, Sgrò P, Salacone P, Caponecchia L, Dondero F, Gandini L. Use of carnitine therapy in selected cases of male factor infertility: a double-blind crossover trial. Fertility and Sterility. 2003. 79(2): 292-300.
- 3 g/day of L-carnitine can increase sperm motility by 40% over three months5 Vitali G, Parente R, Melotti C. Carnitine supplementation in human idiopathic asthenospermia: clinical results. Drugs Under Experimental and Clinical Research. 1995. 21(4): 157-9.
- 2000 mg/day of vitamin C has been shown to increases sperm motility by 39% within two months6 Akmal M, Qadri JQ, Al-Waili NS, Thangal S, Haq A, Saloom KY. Improvement in human semen quality after oral supplementation of vitamin C. Journal of Medicinal Food. 2006. 9(3): 440-2.
- 4 mg/day of the carotene lycopene has been shown to increase sperm motility by 25% in 3 months7 Gupta NP, Kumar R. Lycopene therapy in idiopathic male infertility-a preliminary report. International Urology and Nephrology. 2002. 34(3): 369-72.
- 200 mg selenium combined with 400 IU vitamin E was found to improve motility, morphology or both in 53% of patients over a 14 week trial period8 Moslemi MK, Tavanbakhsh S. Selenium-vitamin E supplementation in infertile men: effects on semen parameters and pregnancy rate. International Journal of General Medicine. 2011. 4:99-104.
- Low levels of vitamin D are associated with poor sperm motility9 Hammoud AO, Meikle AW, Peterson CM, Stanford J, Gibson M, Carrell DT. Association of 25-hydroxy-vitamin D levels with semen and hormonal parameters. Asian Journal of Andrology. 2012. 14(6): 855-9 10 Tartagni M, Matteo M, Baldini D, Tartagni MV, Alrasheed H, De Salvia MA, Loverro G, Montagnani M. Males with low serum levels of vitamin D have lower pregnancy rates when ovulation induction and timed intercourse are used as a treatment for infertile couples: results from a pilot study. Reproductive Biology and Endocrinology. 2015. 13:127.
How do these nutrients work?

Nutrients such as L-carnitine, vitamins E and C, selenium and lycopene are powerful antioxidants. These compounds can help to prevent oxidative stress on cells.
This is important for spermatozoa as they mature. This antioxidant activity can help to protect the developing sperm and ensure healthy motility.
Food enriched with L-carnitine includes lean meats and fish, nuts, seeds, Brussels sprouts, artichokes, asparagus, broccoli, parsley and collard greens.
Sperm Motility Foods
Some of the best sources of vitamin E include kale, almonds, mustard greens, spinach, avocados and sunflower seeds. Citrus fruits are a good source of vitamin C, along with dark leafy greens, berries and tomatoes.
To improve intake of the trace element selenium, men should eat more Brazil nuts, mushrooms, chia seeds, brown rice, seeds and green vegetables.
Lycopene is a bright red carotenoid and found in high concentrations in tomatoes, watermelon, papayas, and red carrots.
Vitamin D is important for sperm cell development. It helps to maintain semen quality and sperm count. The body produces vitamin D following skin exposure to sunlight. If this is insufficient a deficiency in this vitamin may occur.
Improve your sperm motility fertility with micronutrients
Several micronutrients such as vitamins, vitaminoids, amino acids and trace elements have proven themselves effective in improving sperm quantity, mobility and shape. This directly translates into better overall sperm quality and therefore a higher chance of pregnancy.
The benefits of a natural food supplementation therapy for men’ link=” linktarget=” av_uid=’av-fe89e8′[/mfn]
- relatively inexpensive
- effective after three to six months
- able to increase sperm motility by up to 23%, ejaculate volume by up to 33% and sperm count by up to 215%11 Imhof M, Lackner J, Lipovac M, Chedraui P, Riedl C. Improvement of sperm quality after micronutrient supplementation. e-SPEN Journal. 2012. 7(1): e50-e53
- without side effects
For those reasons, male fertility food supplements are most definitely recommended as the first step in the treatment of oligospermia and asthenospermia.
 We have tested the best natural male fertility boosters[/caption[/mfn]
We have tested the best natural male fertility boosters[/caption[/mfn]
Also men who have not yet taken a semen analysis test will benefit from supplementing micronutrients to ensure they are able to deliver high-quality semen.
There are no contraindications or side effects to this form of natural “sperm boosting”.
An excellent and detailed overview of many studies can be found in Steven Sinclair’s Male Infertility: Nutritional and Environmental Considerations.
A considerable range of male fertility supplements available on the UK market.
However, the products differ widely in price and composition. Menfertility.org has compared 10 of them in terms of value for money and the nutrients they provide.
[av_promobox button=’yes’ label=’CLICK ME’ link=’post,47′ link_target=’_blank’ color=’red’ custom_bg=’#444444′ size=’large’ icon_select=’no’ icon=’ue800′ av_uid=’av-dmig8g’[/mfn]
The most effective male fertility nutrients
A multitude of studies has shown that highly dosed nutrients have potentially significant impact on overall sperm quality.
The amino acid L-arginine has been proven to increase sperm count and motility 12 Sinclair S. Male Infertility: Nutritional and Environmental Considerations. Alternative Medicine Review. 2000. 5(1): 28-38 13 Scibona M, Meschini P, Capparelli S, Pecori C, Rossi P, Menchini Fabris GF. L-arginine and male infertility. Italian Journal of Urology and Nephrology. 1994. 46(4):251-3.
Another amino acid L-carnitine has been found to significantly improve sperm concentration (count) and motility over a relatively short period of only 8 weeks 14 Lenzi A, Lombardo F, Sgrò P, Salacone P, Caponecchia L, Dondero F, Gandini L. Use of Carnitine Therapy in Selected Cases of Male Factor Infertility: A Double-Blind Crossover Trial. Fertility and Sterility. 2003. 79(2): 292-300 15 Costa M, Canale D, Filicori M, D’lddio S, Lenzi A. L-carnitine in Idiopathic Asthenozoospermia: A Multicenter Study. Italian Study Group on Carnitine and Male Infertility. Andrologia. 1994. 26(3): 155-9.
Vitamin D has been shown to improve sperm count, motility and morphology16 Jensen MB , Bjerrum PJ, Jessen TE, Nielsen JE, Joensen UN, Olesen IA, Petersen JH, Juul A, Dissing S, Jørgensen N. Vitamin D Is Positively Associated With Sperm Motility and Increases Intracellular Calcium in Human Spermatozoa. Human Reproduction. 2011. 26(6): 1307-17.
Vitamin B9, better known as folic acid has been shown to increase count, motility and morphology 17 Safarinejad MR, Shafiei N, Safarinejad S. Relationship Between Genetic Polymorphisms of Methylenetetrahydrofolate Reductase (C677T, A1298C, and G1793A) as Risk Factors for Idiopathic Male Infertility. Reproductive Sciences. 2011. 18(3): 304-15.
Zinc improves the immune system and significantly improves sperm count in combination with folic acid18 Wong WY, Merkus HMWM, Thomas CMG, Menkveld R, Zielhuis GA, Steegers-Theunissen RPM. Effects of Folic Acid and Zinc Sulfate on Male Factor Subfertility: A Double-Blind, Randomized, Placebo-Controlled Trial. Fertility and Sterility. 2002. 77(3): 491-8.
Selenium in combination with vitamin E has been found to improve motility19 Moslemi MK, Tavanbakhsh S. Selenium-vitamin E Supplementation in Infertile Men: Effects on Semen Parameters and Pregnancy Rate. International Journal of General Medicine. 2011. 4:99-104 20 Keskes-Ammar L, Feki-Chakroun N, Rebai T, Sahnoun Z, Ghozzi H, Hammami S, Zghal K, Fki H, Damak J, Bahloul A. Sperm Oxidative Stress and the Effect of an Oral Vitamin E and Selenium Supplement on Semen Quality in Infertile Men. Archives of Andrology. 2003. 49(2): 83-94 21 Vézina D, Mauffette F, Roberts KD, Bleau G. Selenium-vitamin E Supplementation in Infertile Men. Effects on Semen Parameters and Micronutrient Levels and Distribution. Biological Trace Element Research. 1996. 53(1-3): 65-83.
Why the nutrients must be supplied permanently’ link=” linktarget=” av_uid=’av-clj7uo’[/mfn]
Sperm cells take 11 weeks to mature in the testicles. Only then they are ready for ejaculation.
If you adjust your diet today it will thus take three months for the better sperm to be ready for fertilisation.
You must therefore keep the diet or supplement on an ongoing basis – ideally until your partner is pregnant or you decide for a different treatment.
All of the male fertility supplements in our great test include several of these nutrients at once, albeit at a lower dose. This is a cost-effective and convenient way making this type of fertility therapy affordable and requiring taking only one all-in-one supplement instead of many.
To find out more about the effects of the individual nutrients and how the various supplements compare, please read menfertility.org’s male fertility supplement review.
[av_promobox button=’yes’ label=’CLICK ME’ link=’post,47′ link_target=’_blank’ color=’green’ custom_bg=’#444444′ size=’large’ icon_select=’no’ icon=’ue800′ av_uid=’av-ahkb28′[/mfn]
[av_one_fourth first av_uid=’av-9smvyo’[/mfn][/av_one_fourth[/mfn] [av_one_half av_uid=’av-7is7ps’[/mfn] [av_notification title=’register’ color=’blue’ border=’solid’ custom_bg=’#444444′ size=’medium’ icon_select=’no’ icon=’ue800′ av_uid=’av-k7xs0′[/mfn]
[av_icon_box icon=’ue8d2′ title=’Get your free male fertility food and lifestyle plan with 12 delicious recipes’ link=’manually,https://menfertility.org/register’ linktarget=’_blank’ av_uid=’av-4y1b3k’[/mfn] [av_sidebar widget_area=’Sign-up Widget’ av_uid=’av-2r134g’[/mfn]
[av_one_fourth av_uid=’av-2dp6w0′[/mfn][/av_one_fourth[/mfn]
BIBLIOGRAPHY

Dr. Jones is an experienced consultant in assisted reproduction.
He has worked as a Fertility specialist at Kingston Hospital Assisted Conception and nearly 10 years experience of working in Obstetrics and Gynaecology across hospitals in the UK.
He completed his Masters in Assisted Reproduction Technology and then his PhD, from Imperial College London. Dr. Jones main areas of interest are Single Embryo Transfer, Endometriosis, PCOS and Implantation failure in IVF patients. He is a member of the British Fertility Society and an associate member of the Royal College of Obstetrics and Gynaecology.
Bibliography
- 1La Vignera S, Condorelli RA, Balercia G, Vicari E, Calogero AE. Does alcohol have any effect on male reproductive function? A review of literature. Asian Journal of Andrology. 2013. 15(2): 221-5
- 2Agarwal A, Desai NR, Makker K, Varghese A, Mouradi R, Sabanegh E, Sharma R. Effects of radiofrequency electromagnetic waves (RF-EMW) from cellular phones on human ejaculated semen: an in vitro pilot study. Fertility and Sterility. 2009. 92(4): 1318-25
- 3Gutschi T, Mohamad Al-Ali B, Shamloul R, Pummer K, Trummer H. Impact of cell phone use on men’s semen parameters. Andrologia. 2011. 43(5): 312-6
- 4Lenzi A, Lombardo F, Sgrò P, Salacone P, Caponecchia L, Dondero F, Gandini L. Use of carnitine therapy in selected cases of male factor infertility: a double-blind crossover trial. Fertility and Sterility. 2003. 79(2): 292-300
- 5Vitali G, Parente R, Melotti C. Carnitine supplementation in human idiopathic asthenospermia: clinical results. Drugs Under Experimental and Clinical Research. 1995. 21(4): 157-9
- 6Akmal M, Qadri JQ, Al-Waili NS, Thangal S, Haq A, Saloom KY. Improvement in human semen quality after oral supplementation of vitamin C. Journal of Medicinal Food. 2006. 9(3): 440-2
- 7Gupta NP, Kumar R. Lycopene therapy in idiopathic male infertility-a preliminary report. International Urology and Nephrology. 2002. 34(3): 369-72
- 8Moslemi MK, Tavanbakhsh S. Selenium-vitamin E supplementation in infertile men: effects on semen parameters and pregnancy rate. International Journal of General Medicine. 2011. 4:99-104
- 9Hammoud AO, Meikle AW, Peterson CM, Stanford J, Gibson M, Carrell DT. Association of 25-hydroxy-vitamin D levels with semen and hormonal parameters. Asian Journal of Andrology. 2012. 14(6): 855-9
- 10Tartagni M, Matteo M, Baldini D, Tartagni MV, Alrasheed H, De Salvia MA, Loverro G, Montagnani M. Males with low serum levels of vitamin D have lower pregnancy rates when ovulation induction and timed intercourse are used as a treatment for infertile couples: results from a pilot study. Reproductive Biology and Endocrinology. 2015. 13:127
- 11Imhof M, Lackner J, Lipovac M, Chedraui P, Riedl C. Improvement of sperm quality after micronutrient supplementation. e-SPEN Journal. 2012. 7(1): e50-e53
- 12Sinclair S. Male Infertility: Nutritional and Environmental Considerations. Alternative Medicine Review. 2000. 5(1): 28-38
- 13Scibona M, Meschini P, Capparelli S, Pecori C, Rossi P, Menchini Fabris GF. L-arginine and male infertility. Italian Journal of Urology and Nephrology. 1994. 46(4):251-3
- 14Lenzi A, Lombardo F, Sgrò P, Salacone P, Caponecchia L, Dondero F, Gandini L. Use of Carnitine Therapy in Selected Cases of Male Factor Infertility: A Double-Blind Crossover Trial. Fertility and Sterility. 2003. 79(2): 292-300
- 15Costa M, Canale D, Filicori M, D’lddio S, Lenzi A. L-carnitine in Idiopathic Asthenozoospermia: A Multicenter Study. Italian Study Group on Carnitine and Male Infertility. Andrologia. 1994. 26(3): 155-9
- 16Jensen MB , Bjerrum PJ, Jessen TE, Nielsen JE, Joensen UN, Olesen IA, Petersen JH, Juul A, Dissing S, Jørgensen N. Vitamin D Is Positively Associated With Sperm Motility and Increases Intracellular Calcium in Human Spermatozoa. Human Reproduction. 2011. 26(6): 1307-17
- 17Safarinejad MR, Shafiei N, Safarinejad S. Relationship Between Genetic Polymorphisms of Methylenetetrahydrofolate Reductase (C677T, A1298C, and G1793A) as Risk Factors for Idiopathic Male Infertility. Reproductive Sciences. 2011. 18(3): 304-15
- 18Wong WY, Merkus HMWM, Thomas CMG, Menkveld R, Zielhuis GA, Steegers-Theunissen RPM. Effects of Folic Acid and Zinc Sulfate on Male Factor Subfertility: A Double-Blind, Randomized, Placebo-Controlled Trial. Fertility and Sterility. 2002. 77(3): 491-8
- 19Moslemi MK, Tavanbakhsh S. Selenium-vitamin E Supplementation in Infertile Men: Effects on Semen Parameters and Pregnancy Rate. International Journal of General Medicine. 2011. 4:99-104
- 20Keskes-Ammar L, Feki-Chakroun N, Rebai T, Sahnoun Z, Ghozzi H, Hammami S, Zghal K, Fki H, Damak J, Bahloul A. Sperm Oxidative Stress and the Effect of an Oral Vitamin E and Selenium Supplement on Semen Quality in Infertile Men. Archives of Andrology. 2003. 49(2): 83-94
- 21Vézina D, Mauffette F, Roberts KD, Bleau G. Selenium-vitamin E Supplementation in Infertile Men. Effects on Semen Parameters and Micronutrient Levels and Distribution. Biological Trace Element Research. 1996. 53(1-3): 65-83